General

On Thursday June 25th, the Tager seminar series will continue with a new lecture by Prof. Leila Akkari, who is an Full Professor at Leiden Univeristy, Group Leader in the Tumor Biology and Immunology Department at the Netherlands Cancer Institute (NKI), and Senior Member of the Oncode Institute.
She is a cancer immunologist whose research dissects how macrophage origin, metabolic wiring, and plasticity shape tumor-microenvironment interactions and therapeutic resistance in primary brain and liver cancers. Akkari’s work on macrophages in cancer has been supported by KWF (2017-YIG, 2019, 2021, 2024), NWO Vidi (2019), NWO-XL (2022) and ERC synergy (2024) grants, and is recognized by her integration to the EMBO Young Investigator Program (2021), Board Member of the European Association of Cancer Research (EACR) and by the Ammodo Award 2025 for fundamental research. Dr. Akkari leads an interdisciplinary research team composed of 17 people, spanning from 8 PhD students, 6 post docs and 3 technicians and is deeply committed to mentorship and inclusive scientific culture. She published several pioneering papers on her research, is invited as plenary speaker on several conferences is involved in science communication advocating for equitable training environments and support of early-career researchers, particularly women. Through leadership roles, conference organization, and patient engagement programs within Oncode and NKI, she thrives to foster diversity in scientific representation in STEM.
From Diversity to Dependency: Plasticity and State Evolution of Myeloid Cells in Cancer
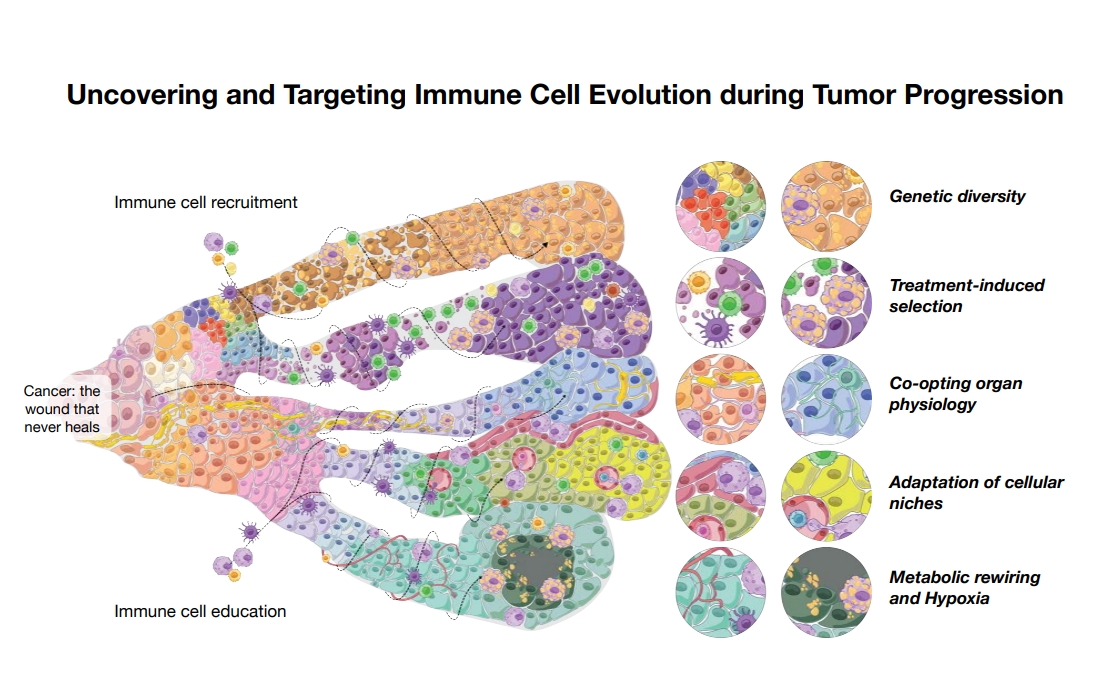
Tumors evolving within chronically damaged and metabolically constrained tissues depend on dynamic metabolic and signaling cross-talk between cancer cells and the immune microenvironment to sustain growth, evade immune control, and acquire therapeutic resistance. Our laboratory interrogates how tissue context, metabolic stress, and immune cell plasticity jointly shape tumor progression and response to therapy. Using integrated multi-omics, spatial profiling, and functional perturbation approaches across preclinical models of brain and liver cancer and complementary patient datasets, we map the spatiotemporal heterogeneity and lineage dynamics of tumor-infiltrating myeloid and lymphoid compartments.
We identify metabolically rewired tumor-associated macrophage, myeloid, and regulatory T cell states that emerge during disease progression and treatment and actively drive immune suppression and resistance to both standard-of-care and immunotherapies. By defining the principles governing immune-metabolic co-evolution in tumors, our work aims to uncover context-specific vulnerabilities that can be leveraged to reprogram the tumor microenvironment and restore durable therapeutic responses.
